41 orbital diagram for copper
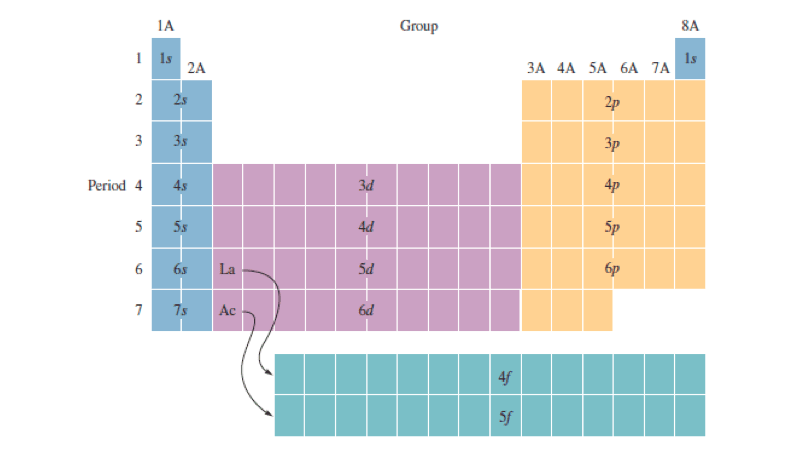
Barium(Ba) electron configuration and orbital diagram Barium(Ba) is the 56th element in the periodic table and its symbol is 'Ba'. This article gives an idea about the electron configuration of barium and orbital diagram, period and groups, valency and valence electrons of barium, bond formation, compound formation, application of different principles. Electron Configuration for Copper (Cu, Cu+, Cu2+) In writing the electron configuration for Copper the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for Copper go in the 2s orbital. The next six electrons will go in the 2p orbital. The p orbital can hold up to six electrons.
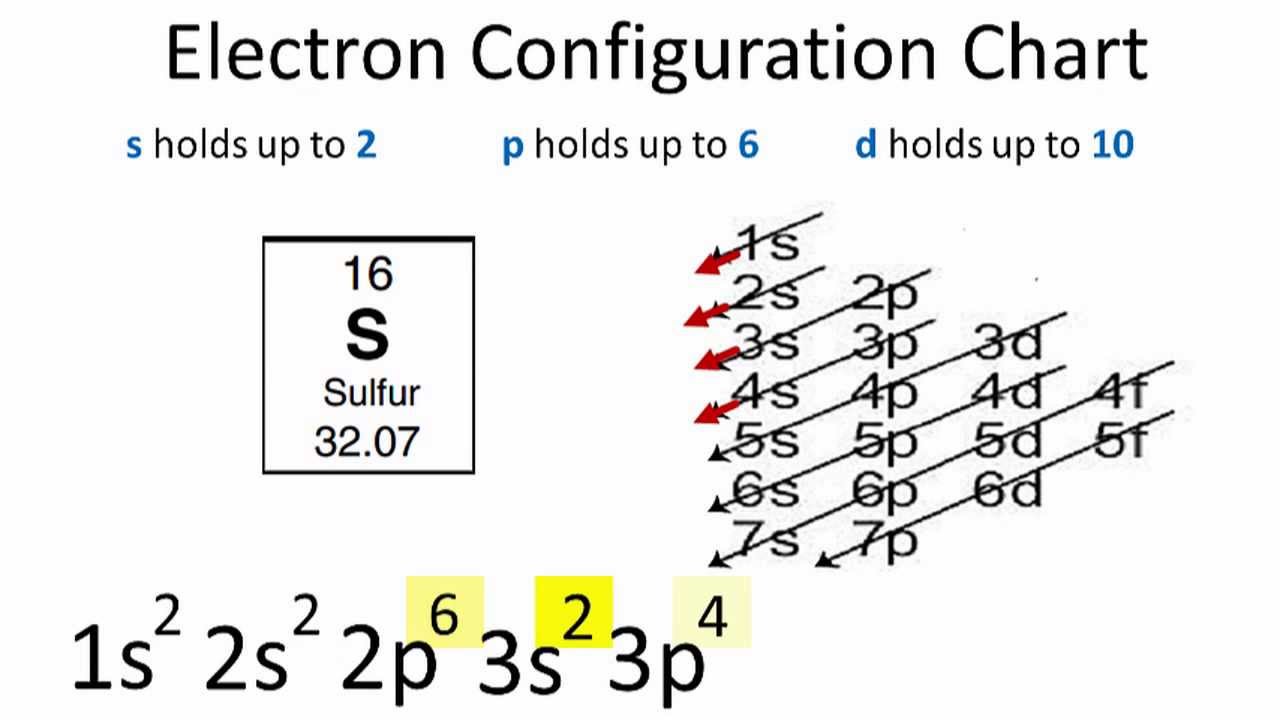
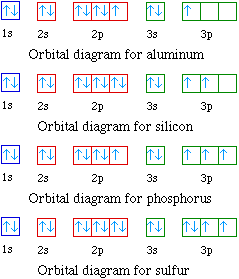
PDF Electron Configurations, Orbital Notation and Quantum Numbers Start by drawing its orbital notation for the outermost, valence electrons. [Ne] ↑↓ ↑↓ ↑ ↑ 3s 3p Sulfur is a nonmetal and tends to gain electrons, creating the -2 charge. Gaining two electrons gives it an octet of 3s23p6. • Copper has two common oxidation states, +1 and +2.

Orbital diagram for copper
DOC Name Draw the orbital diagram for the following elements: Oxygen (O) Titanium (Ti) Silicon (Si) Copper (Cu) For each of the following elements, identify if the electron configuration is correct or incorrect. If it is incorrect, give the fix to the configuration. Carbon (C) = 1s22s22p2. Sulfur (S) = 1s22s22p63p6 Electron configuration for Copper (element 29). Orbital diagram Cu (Copper) is an element with position number 29 in the periodic table. Located in the IV period. Melting point: 1083.5 ℃. Density: 8.92 g/cm 3 . The order of filling the orbitals with electrons in the Cu atom is an exception to the rule. Expected electronic configuration. 1s2 2s2 2p6 3s2 3p6 4s2 3d9. But in reality, one electron moves from ... PDF Molecular Orbital Theory - Octahedral, Tetrahedral or ... The overall molecular orbital energy level diagram for σ-bonding in octahedral complexes can be shown as: Figure 10. The formation of σ-molecular orbitals (bonding, antibonding and non-bonding) in octahedral complexes of transition metals. Buy the complete book with TOC navigation,
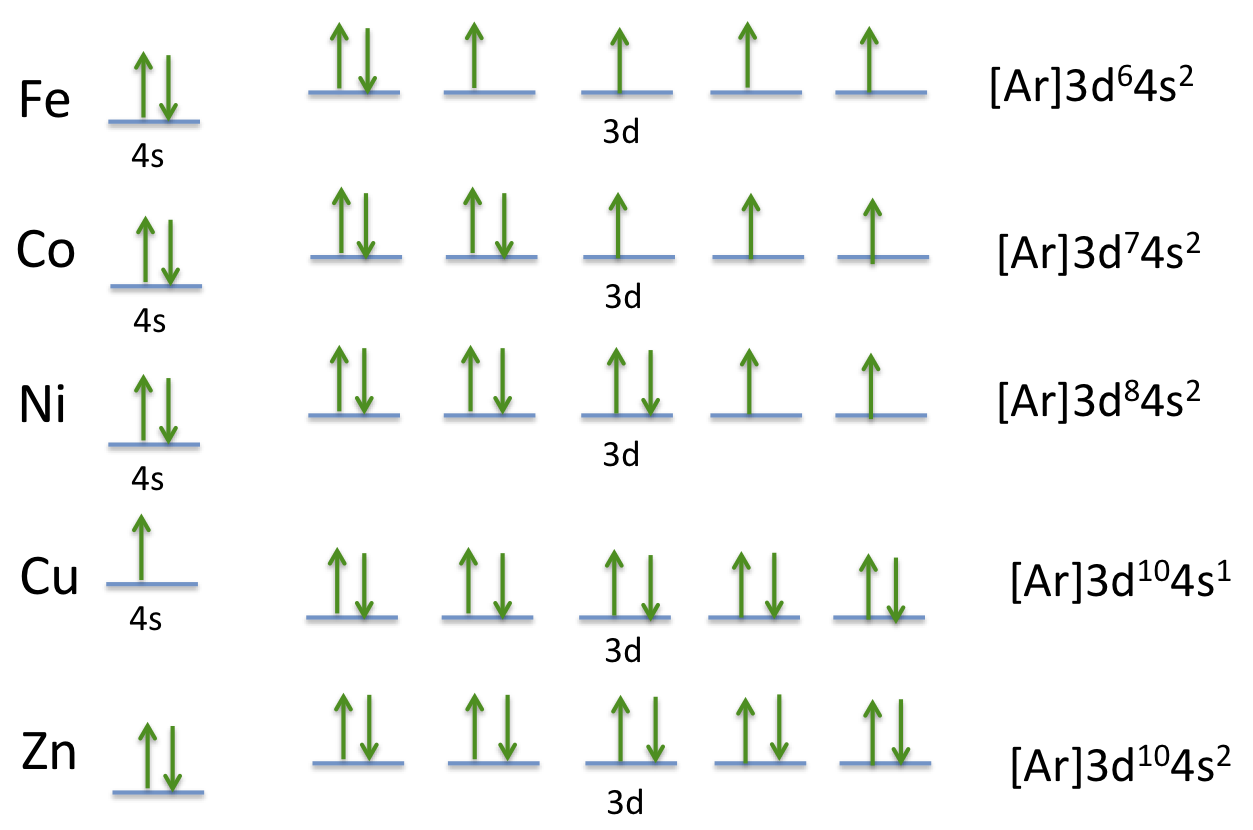
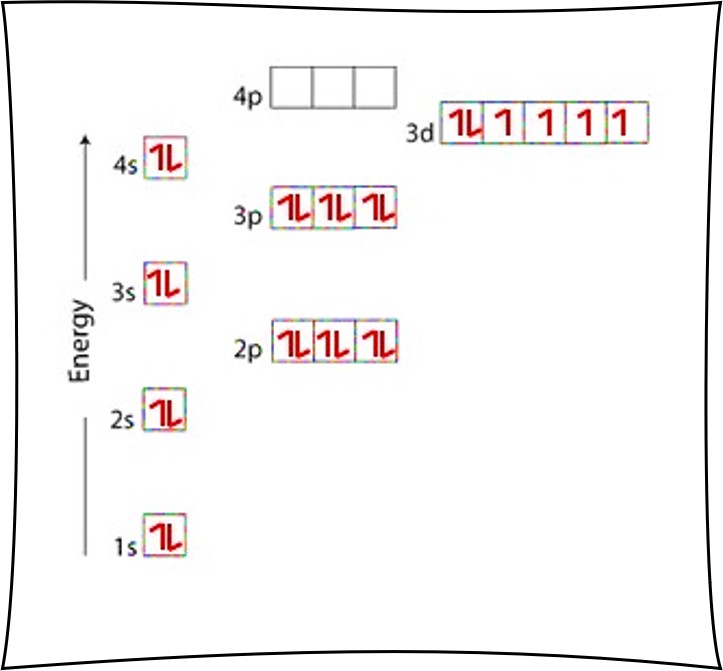
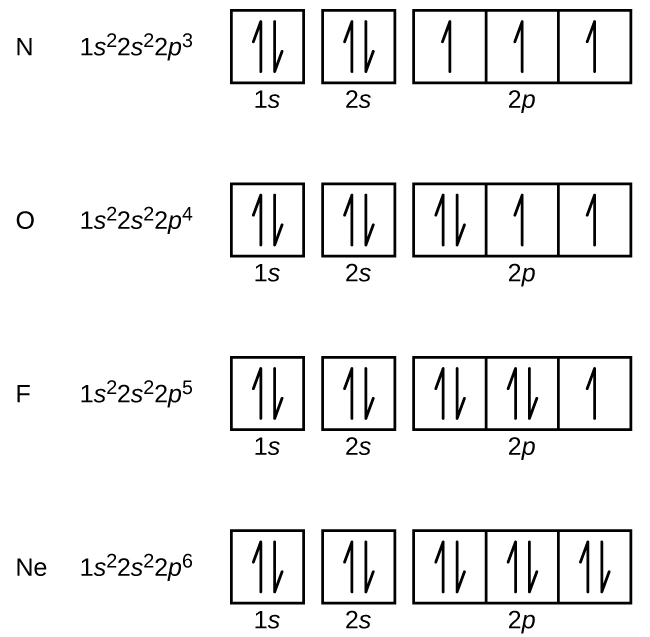
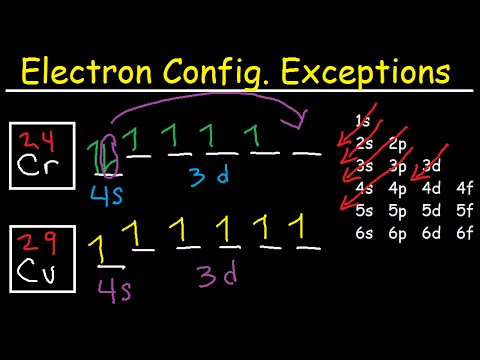
Orbital diagram for copper. Copper, atomic structure - Stock Image - C013/1552 ... Copper (Cu). Diagram of the nuclear composition and electron configuration of an atom of copper-63 (atomic number: 29), the most common isotope of this element. The nucleus consists of 29 protons (red) and 34 neutrons (blue). 29 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). Why are Copper and Chromium exceptions? - CHEMISTRY COMMUNITY Re: Why are Copper and Chromium exceptions? When doing the electron configurations for these elements, they are exceptions to the general rule because a completely full or half full d sub-level is more stable than a partially filled d sub-level, so an electron from the 4s orbital is excited and rises to a 3d orbital. How to Do Orbital Diagrams - Sciencing Electron orbital diagrams and written configurations tell you which orbitals are filled and which are partially filled for any atom. The number of valence electrons impacts on their chemical properties, and the specific ordering and properties of the orbitals are important in physics, so many students have to get to grips with the basics. Copper(Cu) electron configuration and orbital diagram Copper (Cu) orbital diagram 1s is the closest and lowest energy orbital to the nucleus. Therefore, the electron will first enter the 1s orbital. According to Hund's principle, the first electron will enter in the clockwise direction and the next electron will enter the 1s orbital in the anti-clockwise direction.
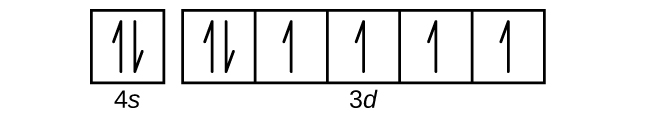
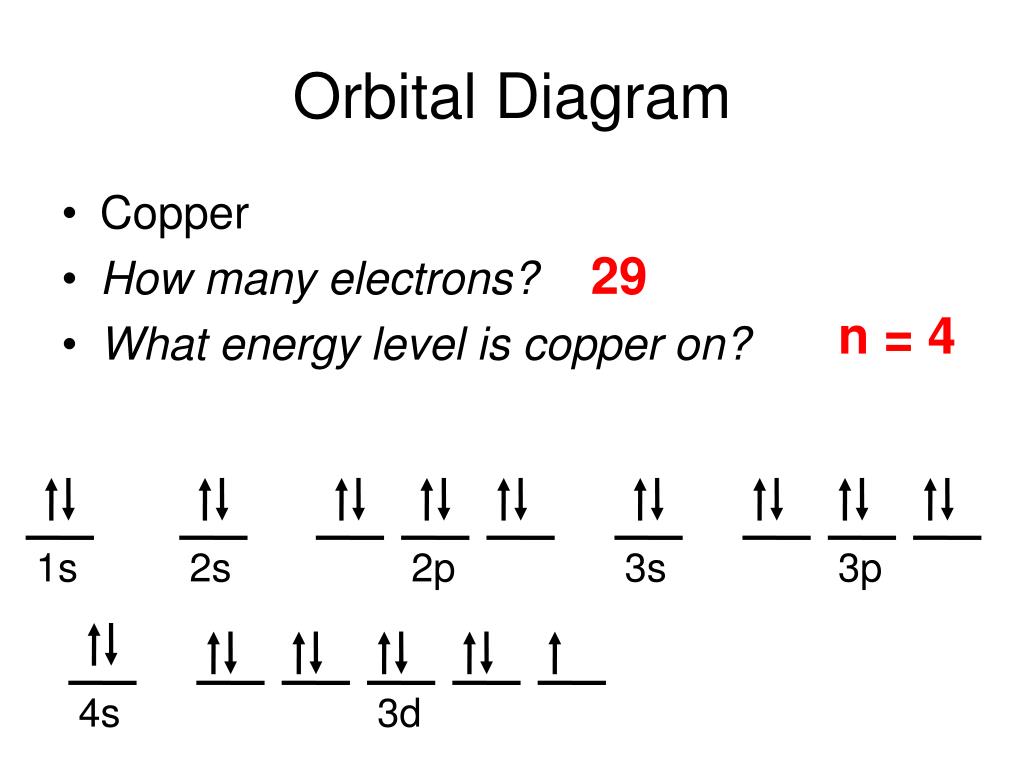
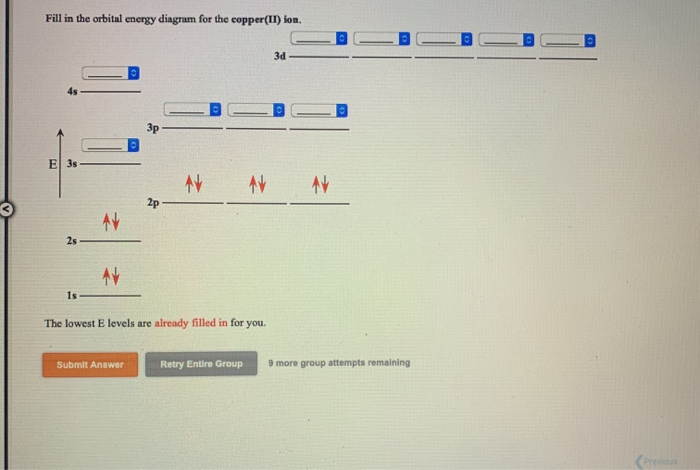
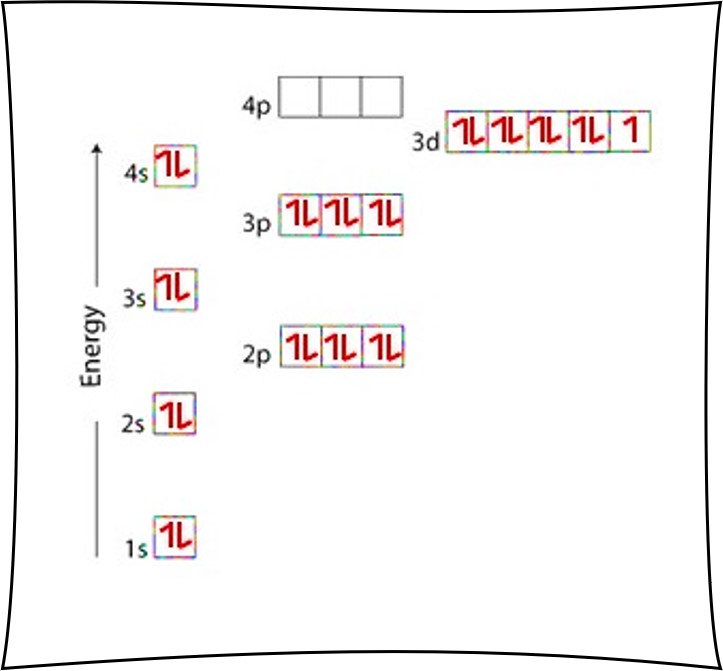
Copper Orbital Diagram - apindustria.padova.it Copper Orbital Diagram 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p. Bromine Orbital Diagram. The sorbital has a spherical shape centered around the origin of the three axes in space. Copper has 29 protons and copper-63 also has (63 - 29) 34 neutrons. Molecular orbital diagram for o2 2. Ion concentration m (mol/kg water). Orbital Diagram of All Elements (Diagrams given Inside) Orbital diagram of Copper (Cu) 30: Orbital diagram of Zinc (Zn) 31: Orbital diagram of Gallium (Ga) 32: Orbital diagram of Germanium (Ge) 33: Orbital diagram of Arsenic (As) 34: Orbital diagram of Selenium (Se) 35: Orbital diagram of Bromine (Br) 36: Orbital diagram of Krypton (Kr) 37: Orbital diagram of Rubidium (Rb) 38: Fill in the orbital energy diagram for cop... | Clutch Prep We’re being asked to fill the orbital energy diagram for copper. Before we can do that, we have to first identify the number of electrons. • In a neutral atom: Atomic number = # of protons = # of electrons. Cu: atomic number = 29 → 29 protons & 29 electrons. • Distribute electrons in the atomic orbitals: 97% (358 ratings) What is the orbital diagram for copper? - Answers Jun 14, 2012 · The electron configuration of copper is 1s22s22p63s23p63d104s1.
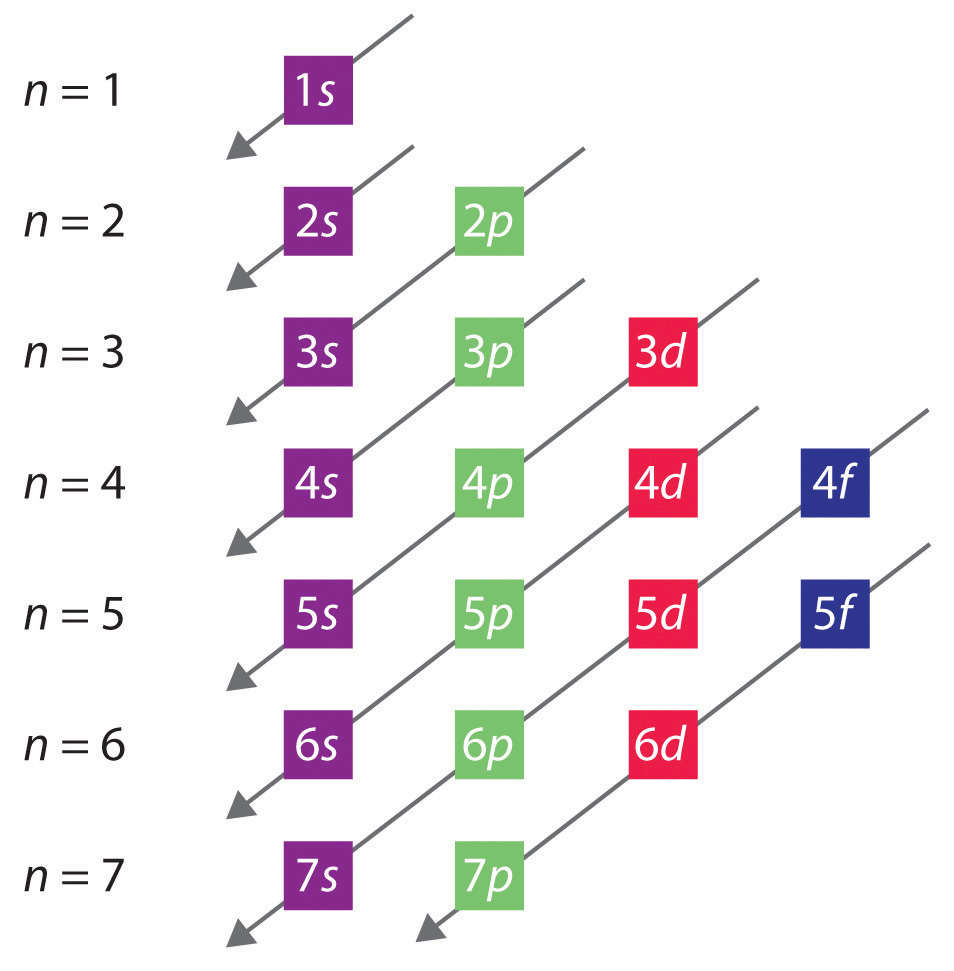
Solved a) Write the full electron configuration for Copper ... Chemistry questions and answers. a) Write the full electron configuration for Copper. b) Write the full orbital diagram for Copper. c) Write the abbreviated electron configuration for Copper. d) How many valence electrons does Copper have? Question: a) Write the full electron configuration for Copper. b) Write the full orbital diagram for Copper. Orbital Diagrams Chemistry Tutorial - AUS-e-TUTE An orbital diagram, or orbital box diagram, is a way of representing the electron configuration of an atom. A box, line, or circle, is drawn to represent each orbital in the electron configuration. (using the Aufau Principle to order the orbitals and hence the boxes, lines or circles, as shown below) 1s. →. 2s. What is the electron configuration of copper? | Socratic However, because the 3d orbital is so much larger then the 4s orbital and the 3d orbital only needs one more electron to be filled, the 3d orbital pulls an electron from the 4s orbital to fill this empty space. This makes the actual electron configuration for copper [Ar] 4s13d10. Electron Configurations Watch later Watch on Answer link PDF Orbital Diagrams, Noble Gas Configuration, Lewis Dot Diagrams Orbital Filling Diagrams •Each box represents an orbital which can hold a max of 2 e- •Aufbau principal -each electron occupies the lowest energy orbital available; German for "build up" •Electrons are notated with an arrow -Up arrow goes first then, down arrow -Arrows represent the opposing spin of electrons 5.2 Quantum Theory & The Atom
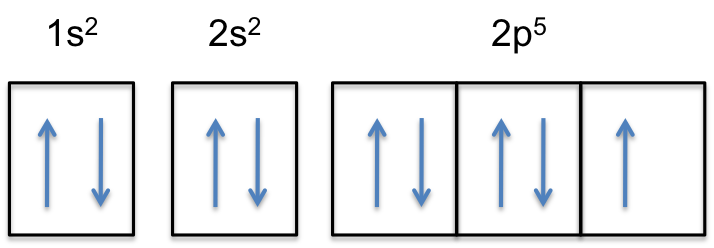
Electron Configurations, Orbital Box Notation (M7Q7 ... The 1s orbital at the bottom of the diagram is the orbital with electrons of lowest energy. The energy increases as we move up to the 2 s and then 2 p , 3 s , and 3 p orbitals, showing that the increasing n value has more influence on energy than the increasing l value for small atoms.
Manganese(Mn) electron configuration and orbital diagram Manganese (Mn) excited state electron configuration and orbital diagram. When a manganese atom is excited, then the manganese atom absorbs energy. As a result, an electron in the 4s orbital jumps to the 4p x sub-orbital. The p-orbital has three sub-orbitals. The sub-orbitals are p x, p y, and p z.
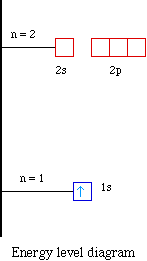
Arrangements of electrons in the orbitals of an atom is ... The orbital diagram, the electron configuration and the energy diagram. All three ways are useful. The next atom is helium with 2 electrons. So the second electron could go into the 1s orbital with the opposite spin of the first electron or it could go into the next orbital in the n = 2 level.
PDF Hybrid Molecular Orbitals - University of Illinois Urbana ... molecular orbital diagram as a non-bonding molecular orbital. 7. There are a total of 6 electrons to add to the molecular orbital diagram, 3 from boron and 1 from each hydrogen atom. sp Hybrid Orbitals in BeH2 1. The Lewis structure shows that the beryllium in BeH 2 makes 2 bonds and has no lone pairs. It is a
What is the orbital diagram for copper? - Answers Definition of orbital diagram? An orbital diagram is used to show how the orbitals of a subshell areoccupied by electrons. The two spin projections are given by arrowspointing up (ms =+1/2) and...
Draw and explain the orbital diagram for copper (Z = 29 ... Answer to: Draw and explain the orbital diagram for copper (Z = 29). By signing up, you'll get thousands of step-by-step solutions to your homework...
Periodic Trends Flashcards - Quizlet Give the ground-state electron configuration for copper (Cu) using noble-gas shorthand. [Ar]3d^104s^1. Item 4: Part A Classify each orbital diagram for ground-state electron configurations by the rule or principle it violates. Aufbau violation:
Copper (Cu) - ChemicalAid Copper (Cu) has an atomic mass of 29. Find out about its chemical and physical properties, states, energy, electrons, oxidation and more.
PDF Molecular Orbital Theory - Octahedral, Tetrahedral or ... The overall molecular orbital energy level diagram for σ-bonding in octahedral complexes can be shown as: Figure 10. The formation of σ-molecular orbitals (bonding, antibonding and non-bonding) in octahedral complexes of transition metals. Buy the complete book with TOC navigation,
Electron configuration for Copper (element 29). Orbital diagram Cu (Copper) is an element with position number 29 in the periodic table. Located in the IV period. Melting point: 1083.5 ℃. Density: 8.92 g/cm 3 . The order of filling the orbitals with electrons in the Cu atom is an exception to the rule. Expected electronic configuration. 1s2 2s2 2p6 3s2 3p6 4s2 3d9. But in reality, one electron moves from ...
DOC Name Draw the orbital diagram for the following elements: Oxygen (O) Titanium (Ti) Silicon (Si) Copper (Cu) For each of the following elements, identify if the electron configuration is correct or incorrect. If it is incorrect, give the fix to the configuration. Carbon (C) = 1s22s22p2. Sulfur (S) = 1s22s22p63p6



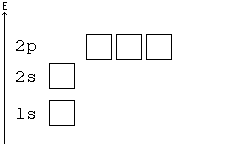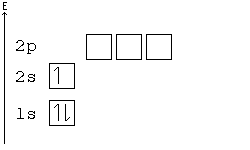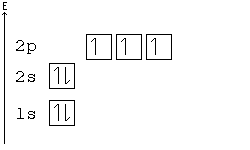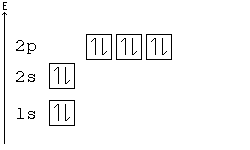













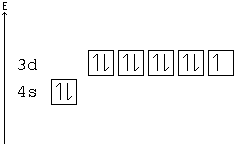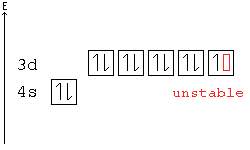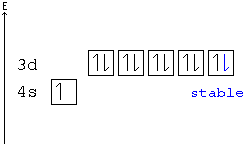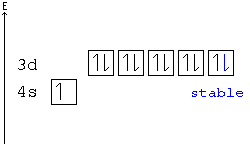
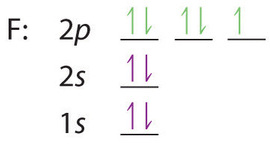



0 Response to "41 orbital diagram for copper"
Post a Comment